
Glia: The Brain’s Forgotten Cells — Friend or Foe?
Mar 08, 2026For decades, neuroscience has told a very neuron-centric story. In fact, you’ve surely heard of them.
Neurons, or nerve cells, are the stars of the show — electrical, excitable, responsible for thought, movement, memory and personality.
But did you know there are other cells in the brain too? A supporting cast called glial cells.
A new 2026 review titled “Glial Cells as Key Mediators in the Pathophysiology of Neurodegenerative Diseases” highlights the different kinds of glial cells within the brain and, importantly, how the extent to which they perform their roles properly may influence neurodegenerative diseases.
Understanding them is changing how we think about — and in the future, even treat — diseases like Alzheimer’s, Parkinson’s, Multiple Sclerosis, Huntington’s and ALS/Motor Neuron Disease.
What Are Glia?
While neurons are responsible for transmitting the electrical signals through the brain and spinal cord, they do not work alone.
In fact, glial cells make up roughly half of the cells in the human brain. In some regions they may even outnumber neurons, although this varies widely.
The word glia comes from the Greek for “glue”, because early anatomists believed they simply held the nervous system together.
Today, we know they do far more.
Glial cells:
- Regulate the chemical environment around neurons
- Protect the neurons against foreign invaders
- Supply metabolic energy
- Maintain electrical insulation (myelin)
- Shape and prune synapses
- Help regulate the blood–brain barrier
In short:
Glia are not passive support cells.
They are active managers of the brain’s internal environment.
The 3 Major Types of Glia
1. Astrocytes — The Environment Managers
Astrocytes are arguably the most versatile cells in the brain.
They are the “environment managers” of neural tissue.
They:
- Regulate neurotransmitters, the signal chemicals between neurons
- Maintain ion and pH (acid-base) balance
- Supply energy to neurons
- Form part of the blood–brain barrier to keep foreign substances or germs out of the brain
- Shape synapses (the “tripartite synapse” model) between neurons
- Coordinate microglia and oligodendrocyte activity
- Regulate water balance
One of their most important roles is glutamate uptake.
Glutamate is the brain’s main excitatory neurotransmitter, essential for overall brain function. However, too much of it leads to excitotoxicity — a state where neurons can become overstimulated and damaged.
Astrocytes prevent this by clearing glutamate from synapses (via the EAAT2/GLT-1 transporter). Remember this, because it comes up quite a bit!.
2. Microglia — The Immune Surveillance Team
Microglia are the brain’s resident immune cells, different to those found in the rest of your body.
They are highly mobile and constantly survey their local environment.
They:
- Consume debris and pathogens in a process called “phagocytosis”
- Prune unnecessary synapses during development
- Release growth factors to help neuroplasticity
- Coordinate inflammatory responses
- Interact with blood vessels and the blood–brain barrier
Importantly, microglia can adopt different activation states.
- M1 – Pro-inflammatory
- M2 – Anti-inflammatory / repair-oriented
However, modern research shows that this is a spectrum of activation, being highly dynamic and context-dependent.
3. Oligodendrocytes — The Insulation & Energy Crew
If neurons are like electrical wires, then oligodendrocytes are the insulation technicians.
Their primary role is to produce myelin — the fatty sheath that wraps around axons (the long “wire” part of a neuron) — allowing electrical signals to travel rapidly and efficiently.
But they also:
- Provide metabolic support to axons
- Work alongside astrocytes to deliver energy substrates like lactate to neurons
- Help maintain long-term axonal survival
- Influence neuroplasticity and repair
- Generate new oligodendrocytes via progenitor cells (OPCs)
Myelination is metabolically expensive, and oligodendrocytes are energy-hungry cells. So when their metabolism fails, axonal conduction and survival suffer.
When functioning normally, this team of glial cells are essential to normal brain function. In-fact, after stroke or traumatic brain injury:
- Microglia help to clear dead tissue and initiate the repair of neurons
- Astrocytes help to stabilise the injured area, and contribute to remodelling
- Oligodendrocyte progenitor (“baby”) cells proliferate and migrate to the damaged area to help remyelinate damaged neurons

Image retrieved from: Bogus, K., Marchesi, N., Campagnoli, L. I. M., Pascale, A., & Pałasz, A. (2026). Glial Cells as Key Mediators in the Pathophysiology of Neurodegenerative Diseases.
When Glia Turn From Friend to Foe
However, the 2026 review focuses on a central point:
Neurodegenerative diseases often involve dysregulated glial responses.
Let’s explore how that plays out, across 5 major neurodegenerative diseases.
This does get a little dense here, as we wanted to maintain accuracy, but feel free to scroll to the section relevant to you (or simply read the bolded summaries in purple!)
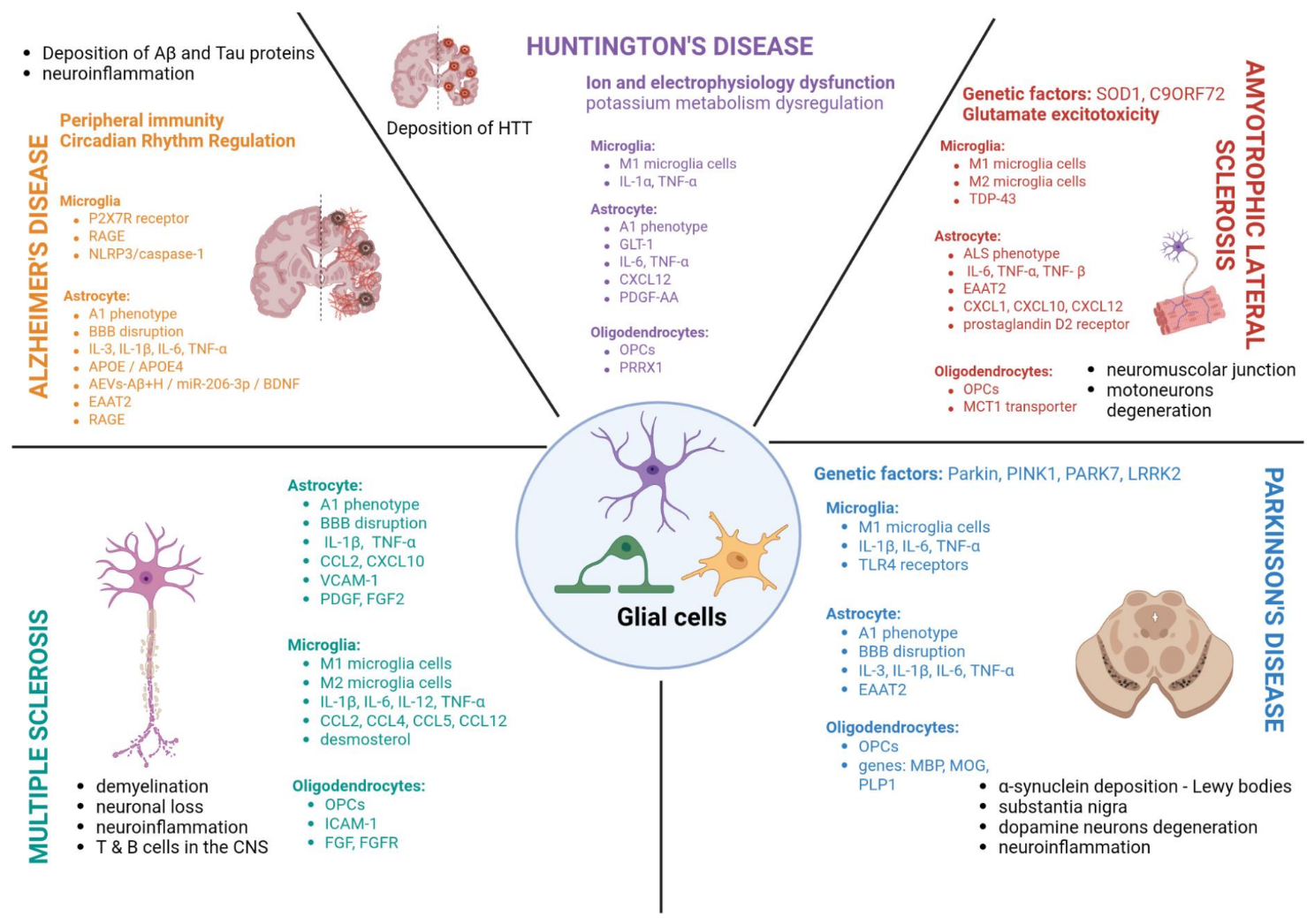
Alzheimer’s Disease (AD)
A key feature of Alzheimer’s are mis-folded abnormal proteins found throughout the brain; namely amyloid-β plaques and tau tangles.
While the exact (likely multifactorial) cause of these proteins and the disease itself remains unclear, neuroinflammation driven by glial cell dysfunction is increasingly recognised as a core component of pathology.
Astrocytes in AD
Astrocytes become “reactive” — changing shape and behaviour — and begin producing inflammatory signals. A key problem in AD is that astrocytes lose some of their ability to clear glutamate, the brain’s main excitatory neurotransmitter we mentioned earlier. When glutamate lingers outside cells, neurons can be overstimulated for prolonged periods, which is harmful.
Inflammatory signals from microglia can push astrocytes into a more damaging state (often labelled “A1”), where they lose supportive functions and contribute to synapse and neuron loss. Astrocyte-driven inflammation can also weaken the blood–brain barrier, making the brain’s protective boundary more permeable to outside substances.
Microglia in AD
Early in the disease, microglia attempt to help by surrounding and clearing amyloid deposits. With ongoing exposure and ageing, many become stuck in a long-term alarm state. They release inflammatory chemicals and reactive oxygen species (ROS), which damage nearby synapses and neurons. At the same time, they become less effective at clearing amyloid, allowing inflammation and protein buildup to reinforce each other.
Oligodendrocytes in AD
This review focuses more on microglia and astrocytes in AD. It still highlights that chronic inflammation and oxidative stress can damage cell energy systems (mitochondria), reducing the brain’s overall support for neurons over time — including the systems involved in maintaining healthy neural wiring.
📌Alzheimer's Summary:
In Alzheimer’s, glial cells initially attempt to protect the brain — clearing amyloid and stabilising the environment — but over time chronic activation, impaired glutamate regulation, and sustained inflammation accelerate neuronal damage. The disease is not just protein buildup, but a breakdown of the brain’s immune and metabolic support system.
Parkinson’s Disease (PD)
Parkinson’s involves degeneration of neurons in a deep part of the brain called the substantia nigra — specifically those responsible for producing dopamine, a key neurotransmitter involved in movement, motivation, pleasure and reward. It is also associated with the accumulation of misfolded α-synuclein protein, forming “Lewy bodies”.
Astrocytes in PD
In PD, astrocytes can react to both α-synuclein deposits and the inflammatory chemicals released by microglia. In response, they may contribute further to the inflammatory environment by releasing their own signalling molecules. A key issue highlighted in the review is reduced glutamate uptake (linked to lower EAAT2 expression), which can leave excess glutamate in brain tissue and increase the risk of long-term overstimulation and neuronal damage.
Astrocytes may also attempt to capture and clear α-synuclein from outside neurons. However, with ongoing stress and inflammation, their ability to regulate the environment becomes impaired.
Microglia in PD
Microglia detect Lewy bodies as abnormal material, triggering activation. Once activated, they release inflammatory chemicals and nitric oxide, and over time can shift toward a more damaging inflammatory state. Brain scans and post-mortem studies show evidence of persistent microglial activation in key PD regions. Ongoing inflammation may also disrupt the blood–brain barrier, allowing further immune activity within the brain.
Oligodendrocytes in PD
The review describes emerging evidence that α-synuclein can accumulate in oligodendrocytes, affecting both myelin production and the metabolic support that axons rely on. It also notes disrupted development of oligodendrocyte precursor cells (OPCs) and early reductions in myelin-related genes. These changes may contribute to alterations in neural “wiring” and symptom progression over time.
📌Parkinson's Summary:
In Parkinson’s disease, misfolded α-synuclein doesn’t just damage neurons directly — it triggers prolonged glial activation. Over time, inflammatory signalling, impaired glutamate balance, and disrupted metabolic support contribute to the vulnerability of dopamine-producing cells.
Multiple Sclerosis (MS)
Multiple sclerosis is an autoimmune disease, where the body’s own immune system attacks the central nervous system. Specifically, the myelin sheath and axon bodies of neurons are broken down in a process called “demyelination”.
But as you now know, oligodendrocytes are responsible for maintaining myelin. So what is going wrong?
Astrocytes in MS
Astrocytes play a major role in blood–brain barrier disruption in MS. When activated, they release signalling molecules that increase barrier permeability, making it easier for immune cells to enter the brain and spinal cord.
Astrocytes can also influence whether repair occurs. In some contexts, they release factors that contribute to scar formation and limit regeneration. In others, they help coordinate repair by clearing debris, recruiting OPCs, and supporting the survival of newly forming oligodendrocytes. Their role depends heavily on the surrounding inflammatory environment.
Microglia in MS
Microglia have a dual role in MS. In active lesions, they contribute to damage by releasing inflammatory chemicals and engulfing myelin. However, they are also essential for repair. Clearing myelin debris is a necessary first step before new myelin can form.
Regions of successful remyelination tend to show more repair-supporting microglial states (often labelled “M2”). The timing and balance of these states strongly influence whether recovery occurs.
Oligodendrocytes in MS
Oligodendrocytes are directly targeted in MS, leading to demyelination and slowed signal conduction. The brain attempts to repair this damage using oligodendrocyte precursor cells (OPCs), but chronic inflammation and oxidative stress can prevent these precursor cells from maturing into fully functioning myelin-producing cells. Successful remyelination often depends on the local inflammatory environment settling down.
📌MS Summary:
In MS, glial cells sit at the centre of both damage and repair. Microglia and astrocytes can drive inflammation and demyelination — yet they are also essential for clearing debris and enabling remyelination, depending on the surrounding inflammatory environment.
Huntington's Disease (HD)
Huntington’s disease is caused by a genetic mutation in the huntingtin gene, which produces a protein essential for healthy neuron function.
Although traditionally viewed as a neuronal disorder, glial cells are now considered early and active contributors to disease progression.
Astrocytes in HD
Astrocytes in HD can lose key supportive functions early in the disease process. One of the most important changes is reduced expression of GLT-1, a transporter responsible for clearing glutamate. When glutamate accumulates, neurons are exposed to prolonged overstimulation, increasing vulnerability.
These functional changes can occur before obvious cell loss, meaning the system may be under strain long before structural damage becomes visible. Experimental studies suggest that improving astrocyte electrical stability and glutamate regulation can partially restore circuit function in models of HD.
Microglia in HD
Microglia contribute to disease progression by sustaining inflammation and influencing astrocyte behaviour. Inflammatory microglia release signalling molecules such as TNF-α, IL-1α and C1q, which can convert astrocytes into a neurotoxic “A1” state. This microglia–astrocyte interaction worsens the environment for neurons and accelerates degeneration.
Oligodendrocytes in HD
The review discusses oligodendrocytes as part of the broader glial involvement in HD, although in less detail than astrocytes and microglia. The central idea is that multiple glial types can both lose protective functions and adopt harmful behaviours early in the disease process.
📌Huntington's Summary:
In Huntington’s disease, glial dysfunction appears early — often before visible neuron loss. Loss of astrocyte glutamate regulation and harmful microglia–astrocyte interactions create an environment that accelerates circuit instability and neuronal degeneration.
Amyotrophic Lateral Sclerosis (ALS)
ALS involves degeneration of motor neurons responsible for transmitting movement signals in both the central (brain and spinal cord) and peripheral nervous system. In Australia and some other countries, ALS is commonly referred to as Motor Neuron Disease (MND).
However, despite the name, glial cells may be critical drivers of the disease’s progression.
Astrocytes in ALS
Astrocytes in ALS can lose their ability to clear glutamate due to reduced EAAT2 expression. Excess glutamate leads to overstimulation and disruption of motor neuron function, a process known as excitotoxicity.
Astrocytes may also release inflammatory and toxic soluble factors that directly harm motor neurons. Laboratory and animal studies show that ALS astrocytes can shrink motor neuron cell bodies and damage axons.
Microglia in ALS
Microglia are consistently activated in ALS, particularly around injured motor neurons. The review describes a shift over time: early stages may show more protective microglial behaviour, while later stages are characterised by stronger pro-inflammatory activity (often labelled “M1”), increased oxidative stress, and greater neuronal injury. These later microglial states are associated with faster disease progression.
Oligodendrocytes in ALS
Oligodendrocyte dysfunction can occur early in ALS, sometimes before clear motor neuron death. Although precursor cells may proliferate, they often fail to mature properly and replace lost oligodendrocytes. This contributes to myelin disruption.
Oligodendrocytes normally provide metabolic support to motor neurons by delivering fuel such as lactate through a transporter called MCT1. When this support system fails, motor neurons become more vulnerable to degeneration.
📌ALS Summary:
In ALS, motor neuron degeneration is strongly influenced by glial failure. Reduced glutamate clearance, chronic microglial activation, and impaired metabolic support together create a hostile environment that drives disease progression.
 Across these conditions, a clear pattern emerges: glial cells shift from regulated support to chronic dysregulation. Inflammation persists, metabolic balance falters, and neurons lose the stability they depend on.
Across these conditions, a clear pattern emerges: glial cells shift from regulated support to chronic dysregulation. Inflammation persists, metabolic balance falters, and neurons lose the stability they depend on.
The Big Takeaway.
So clearly glial cells are not background players.
When performing their roles properly, they are regulators of:
- Homeostasis
- Energy metabolism
- Immune balance
- Synaptic stability
- As well as structural integrity
And by contrast, neurodegenerative diseases are not simply diseases of neurons, but of the brain’s whole support ecosystem.
Microglia become chronically activated.
Astrocytes lose regulatory balance.
Oligodendrocytes fail to maintain insulation and energy supply.
And when the support network collapses, neurons cannot survive.
Where Treatment May Be Heading
Now we’ve spent a lot of time looking at how glial cell function can breakdown, and how this contributes to various diseases.
However there is good news! This better understanding of how the brain functions as a whole, not just via neurons, provides a key avenue by which new medical treatments can be discovered.
In fact, the review highlights growing interest in:
- Targeting microglial receptors (e.g., P2X7, NLRP3)
- Modulating inflammatory cascades
- Restoring glial metabolic function
- Immunotherapies aimed at balancing glial phenotypes (variations of expression)
The future of therapy for many of these diseases may involve restoring equilibrium in the brain’s ecosystem — not just targeting neurons directly as many traditional approaches have done.
Beyond the Review: Lifestyle & Glial Health
Now that you have a better understanding of glial cells, we want to branch outside the findings of this paper slightly.
There is known and emerging evidence that certain lifestyle factors, while not responsible for preventing disease alone, can significantly improve glial cell (and by extension, brain) health!
For example:
- Exercise may promote anti-inflammatory microglial states and increases BDNF (a growth-supporting protein).
- Sleep supports astrocytic housekeeping and metabolic clearance.
- Chronic stress activates inflammatory pathways that dysregulate glia.
- Insulin resistance and poor diet amplify oxidative stress and inflammatory signalling.
The brain functions as an interconnected system. Supporting that system — through research, clinical care and lifestyle — matters.
So with that in mind, stay tuned for future blogposts and PhysiKit courses as we continue unpacking the science behind neurological disease and how it translates into practical strategies for brain health.
And if you haven't already, make sure to listen to our NeuroNotes Podcast episode 6 where we cover this same paper!
References
Bogus, K., Marchesi, N., Campagnoli, L. I. M., Pascale, A., & Pałasz, A. (2026). Glial Cells as Key Mediators in the Pathophysiology of Neurodegenerative Diseases. International Journal of Molecular Sciences, 27(2), 884. https://doi.org/10.3390/ijms27020884
Stay Informed!
News, updates, and science delivered to your inbox.
We will never sell your information, for any reason.

